Protein scaffold dna4/30/2023  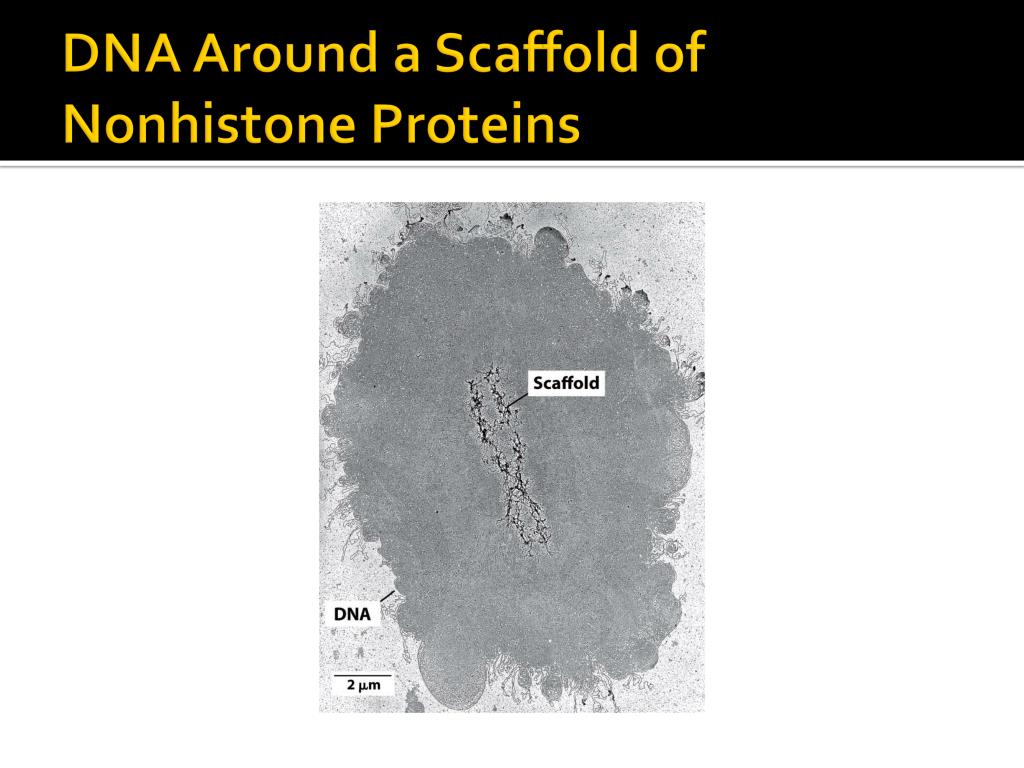
However, its involvement in maintaining chromosomal order in vivo is still controversial. Much evidence has been presented on the influence of this scaffold on the regulation of DNA replication, gene expression and RNA processing (reviewed in Nickerson et al., 1995). Indeed, a proteinaceous framework, termed nuclear matrix or scaffold, can be isolated from the nuclei of nearly all cell types and organisms (reviewed in Berezney et al., 1995). One much debated possibility is that the internal order of the nucleus is determined by a specific attachment of chromatin to an underlying skeleton ( Gasser and Laemmli, 1987). However, even the most basic principles of internal nuclear architecture are not yet fully understood. Whole chromosomes ( Lichter et al., 1988) and even single genetic loci ( Marshall et al., 1996) occupy well-defined areas in the interphase nucleus, and all processes of RNA metabolism are precisely located in distinct nuclear regions ( Xing et al., 1993). In recent years, it has become increasingly clear that the nucleus of eukaryotic cells is a highly ordered organelle. Our results suggest that detachment of SAF–A from SARs, caused by apoptotic proteolysis of its DNA-binding domain, is linked to the formation of oligonucleosomal-sized DNA fragments and could therefore contribute to nuclear breakdown in apoptotic cells. On the other hand, cleavage does not compromise the association of SAF-A with hnRNP complexes, indicating that the function of SAF-A in RNA metabolism is not affected in apoptosis. Cleavage occurs within the bipartite DNA-binding domain, resulting in a loss of DNA-binding activity and a concomitant detachment of SAF–A from nuclear structural sites. During apoptosis, but not during necrosis, SAF-A is cleaved in a caspase-dependent way. We now show that the protein has a novel scaffold-associated region (SAR)-specific bipartite DNA-binding domain which is independent from the previously identified RNA-binding domain, the RGG box. Evidence from previous experiments had suggested that SAF-A most likely has at least two different functions, being involved both in nuclear architecture and RNA metabolism. The scaffold attachment factor A (SAF-A) is an abundant component of the nuclear scaffold and of chromatin, and also occurs in heterogeneous nuclear ribonucleoprotein (hnRNP) complexes. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
